
Patient Brochure
An overview of Tivdak and what to expect from treatment
What is TIVDAK? TIVDAK is a prescription medicine used to treat adults with cervical cancer that has returned or has spread to other parts of the body, and who have received chemotherapy that did not work or is no longer working.
Select the resources you would like to send via email
0 selected
Send by Email

Patient Brochure
An overview of Tivdak and what to expect from treatment


Caregiver Brochure
A resource providing guidance to caregivers about supporting their loved ones on Tivdak


TivdakTexts Brochure
A guide to a text message support program designed to assist you throughout treatment


Important Facts
A short summary of key information about Tivdak
SEAGEN SECURE®

Seagen Secure® is a personalized support program for people prescribed Tivdak. When you or your caregiver calls Seagen Secure®, you will speak with an Oncology Access Advocate who can answer your questions and provide information about available resources and support.†
*Eligibility criteria apply. Financial support may be provided through third-party organizations that have their own eligibility criteria and enrollment processes. Pfizer does not guarantee that enrollment will result in assistance and/or reimbursement.
†Information provided by an Oncology Access Advocate is not intended to be a substitute for clinical advice from your healthcare provider. Discuss any questions you may have about your disease and your treatment with your healthcare team.
‡Seagen Secure® programs available for qualifying patients. Pfizer does not guarantee that enrollment will result in coverage, reimbursement, or financial support.
TivdakTexts
TivdakTexts is a free text message support program designed to assist you throughout treatment§
Some benefits of TivdakTexts include:
Infusion reminders
Eye drop reminders
Inspirational messages
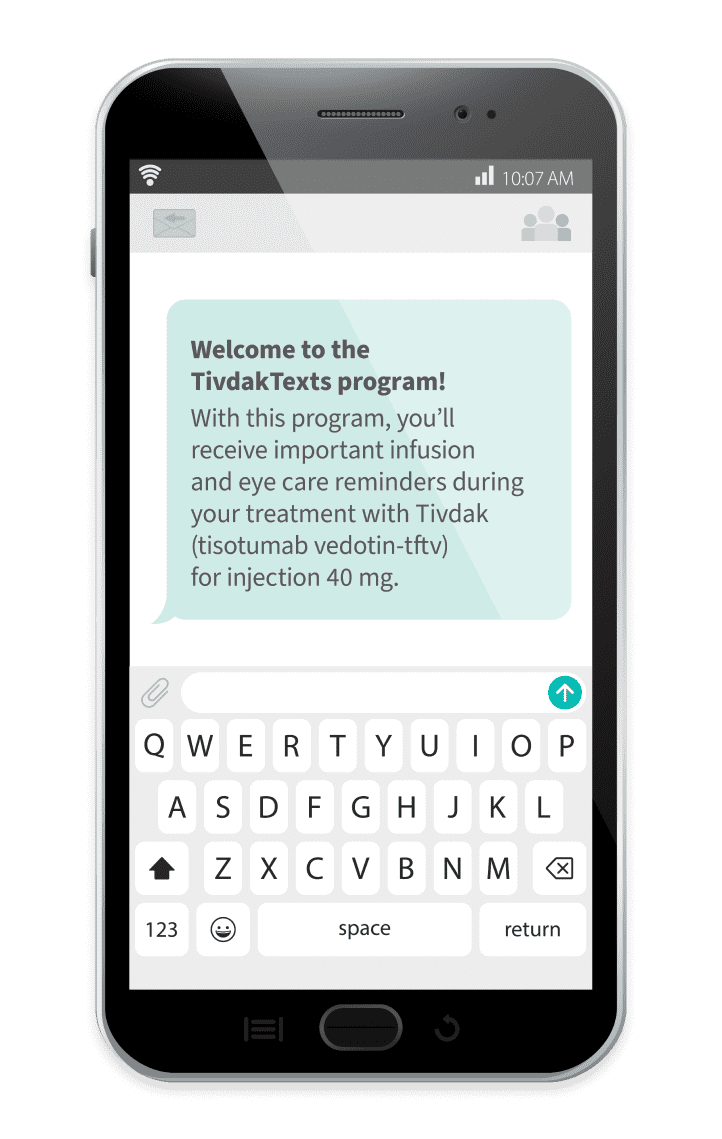
To sign up for TivdakTexts, you can follow these steps:
On your mobile phone, text the word JOIN to 74277
When you get a message back from us, text back AGREE to confirm your participation in the support program
Then provide the following information:
§Message and data rates may apply. Check with your mobile service provider. Message frequency will be based on your selections. Text HELP for help with the program. Text STOP to stop receiving messages. See Terms of Use and Conditions. By initiating a text message, you agree to receive automated text messages from Pfizer. You can unsubscribe at any time. You represent that you are the subscriber of the number provided and 18 or older. You must notify Pfizer immediately if you relinquish the number provided. Consent is not required as a condition of purchasing any products or services.
Find an eye specialist
An eye specialist will help monitor your eye health throughout treatment. Use this locator tool to find one near you.
Advocacy groups
When you have advanced cancer, it can be hard to know where to turn. There are a number of groups offering care and support throughout your experience.
These patient support groups are included as a reference for patients and caregivers. Their inclusion does not imply endorsement from the individual patient support groups listed.

Cancer Support Community
A worldwide network committed to ensuring that no one faces cancer alone by providing navigation, support, and resources through face-to-face, telephone, and online programming.

Cervivor
A cervical cancer awareness and support community, a learning tool, an advocacy resource, and an online retreat for healing, connecting, and thriving.

Family Reach
Family Reach delivers financial education, financial coaching, resource navigation, and emergency relief to patients and caregivers facing a cancer diagnosis.

Foundation for Women’s Cancer
A foundation dedicated to increasing research, education, and awareness about gynecologic cancer risk, prevention, early detection, and optimal treatment.

National Cervical Cancer Coalition
A grassroots organization that helps women, family members, and caregivers battle the personal issues related to cervical cancer and HPV, and advocates for cervical health in all women.

Triage Cancer
A nonprofit group that provides education on the practical and legal issues that may impact individuals diagnosed with cancer and their caregivers through free events, materials, and resources.
By clicking these links, you will be redirected to websites that are neither owned nor controlled by Pfizer or Genmab. Pfizer and Genmab are not responsible for the content or services of these sites.
Hear a real Tivdak PATIENT’S STORY

What to expect DURING TREATMENT

See the STUDY RESULTS

Eye problems are common with TIVDAK and can be severe. TIVDAK can cause changes to the surface of your eye that can lead to dry eyes, eye redness, eye irritation, corneal ulcers, blurred vision, and severe vision loss. Tell your healthcare provider if you develop new or worsening vision changes or eye problems during treatment.
Your healthcare provider will send you to an eye specialist to check your eyes before you start treatment with TIVDAK, before each infusion for your first 9 infusions of TIVDAK, and as needed for any new or worsening signs or symptoms of eye problems. Your healthcare provider will ask if you have any signs or symptoms of eye problems before each infusion. You will be referred to an eye specialist for any new or worsening signs or symptoms of eye problems.
Your healthcare provider will prescribe 3 different types of eye drops before you start treatment with TIVDAK. Bring the eye drops with you to each infusion and use them as directed by your healthcare provider to reduce your risk of eye problems:
Do not wear contact lenses throughout your treatment with TIVDAK unless you are told to use them by your eye specialist.
Serious side effects of TIVDAK may include:
Eye problems. See “What is the most important information I should know about TIVDAK?”
Nerve problems (peripheral neuropathy) are common with TIVDAK and can be serious. Tell your healthcare provider right away if you have new or worsening numbness or tingling in your hands or feet or muscle weakness.
Bleeding problems (hemorrhage) are common with TIVDAK and can be serious. Tell your healthcare provider or get medical help right away if you have signs or symptoms of bleeding during treatment with TIVDAK, including blood in your stools or black stools (looks like tar), blood in your urine, cough up or vomit blood, unusual vaginal bleeding, or any unusual or heavy bleeding.
Lung problems. TIVDAK may cause severe or life-threatening inflammation of the lungs that can lead to death. Tell your healthcare provider right away if you have new or worsening symptoms, including trouble breathing, shortness of breath, or cough.
Severe skin reactions. TIVDAK may cause severe or life-threatening skin reactions that can lead to death. Tell your healthcare provider or get medical help right away if you have signs or symptoms of a severe skin reaction during treatment with TIVDAK, including:
The most common side effects of TIVDAK include:
Your healthcare provider may decrease your dose of TIVDAK, temporarily stop, or completely stop treatment if you have side effects.
Tell your healthcare provider about all your medical conditions, including if you:
are breastfeeding (nursing) or plan to breastfeed. It is not known if TIVDAK passes into your breast milk. Do not breastfeed during treatment and for at least 3 weeks after the last dose of TIVDAK
Females who can become pregnant:
Males with female partners who can become pregnant:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking TIVDAK with certain other medicines may cause side effects.
These are not all of the possible side effects of TIVDAK. Discuss side effects with your healthcare provider. You may report side effects to FDA at 1-800-FDA-1088.
Please see the full Prescribing Information with Medication Guide about TIVDAK including an IMPORTANT WARNING.
TIVDAK is a prescription medicine used to treat adults with cervical cancer:
Eye problems are common with TIVDAK and can be severe. TIVDAK can cause changes to the surface of your eye that can lead to dry eyes, eye redness, eye irritation, corneal ulcers, blurred vision, and severe vision loss. Tell your healthcare provider if you develop new or worsening vision changes or eye problems during treatment.
Your healthcare provider will send you to an eye specialist to check your eyes before you start treatment with TIVDAK, before each infusion for your first 9 infusions of TIVDAK, and as needed for any new or worsening signs or symptoms of eye problems. Your healthcare provider will ask if you have any signs or symptoms of eye problems before each infusion. You will be referred to an eye specialist for any new or worsening signs or symptoms of eye problems.
Your healthcare provider will prescribe 3 different types of eye drops before you start treatment with TIVDAK. Bring the eye drops with you to each infusion and use them as directed by your healthcare provider to reduce your risk of eye problems:
Do not wear contact lenses throughout your treatment with TIVDAK unless you are told to use them by your eye specialist.
Serious side effects of TIVDAK may include:
Eye problems. See “What is the most important information I should know about TIVDAK?”
Nerve problems (peripheral neuropathy) are common with TIVDAK and can be serious. Tell your healthcare provider right away if you have new or worsening numbness or tingling in your hands or feet or muscle weakness.
Bleeding problems (hemorrhage) are common with TIVDAK and can be serious. Tell your healthcare provider or get medical help right away if you have signs or symptoms of bleeding during treatment with TIVDAK, including blood in your stools or black stools (looks like tar), blood in your urine, cough up or vomit blood, unusual vaginal bleeding, or any unusual or heavy bleeding.
Lung problems. TIVDAK may cause severe or life-threatening inflammation of the lungs that can lead to death. Tell your healthcare provider right away if you have new or worsening symptoms, including trouble breathing, shortness of breath, or cough.
Severe skin reactions. TIVDAK may cause severe or life-threatening skin reactions that can lead to death. Tell your healthcare provider or get medical help right away if you have signs or symptoms of a severe skin reaction during treatment with TIVDAK, including:
The most common side effects of TIVDAK include:
Your healthcare provider may decrease your dose of TIVDAK, temporarily stop, or completely stop treatment if you have side effects.
Tell your healthcare provider about all your medical conditions, including if you:
are breastfeeding (nursing) or plan to breastfeed. It is not known if TIVDAK passes into your breast milk. Do not breastfeed during treatment and for at least 3 weeks after the last dose of TIVDAK
Females who can become pregnant:
Males with female partners who can become pregnant:
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking TIVDAK with certain other medicines may cause side effects.
These are not all of the possible side effects of TIVDAK. Discuss side effects with your healthcare provider. You may report side effects to FDA at 1-800-FDA-1088.
Please see the full Prescribing Information with Medication Guide about TIVDAK including an IMPORTANT WARNING.
TIVDAK is a prescription medicine used to treat adults with cervical cancer:
By clicking this link, you will be redirected to a website that is neither owned nor controlled by Pfizer or Genmab. Pfizer and Genmab are not responsible for the content or services of this site.
Please select 1 or more resources.